|
The NF3 molecule has a trigonal pyramidal geometry shape because it. Going back to your question, we are supposed to compare $\ce$ also. The F-N-F bond angle is 97 degrees in the trigonal pyramidal NF3 molecular geometry. You can think of how s orbitals are spherical in shape, and a large s character would lead to more "spherical" hybrid orbitals, and that would lead to larger bond angles. The shape is distorted because of the lone pairs of electrons. What has all of this got to do with bond angles? Well, more s character leads to larger bond angles. It has a molecular geometry of trigonal pyramidal which also looks like a distorted tetrahedral structure. In the NF3 molecule, nitrogen is attached to three fluorine atoms via a single bond and has a molecular weight of 71.00 g/mol. Thus, Bent's rule can just as easily be formulated as follows: p character concentrates in orbitals directed towards electronegative substituents (the central atom doesn't have to waste its low energy s orbitals, the orbitals on the electronegative atom, can (possibly) take care of it. Which is why molecules like to use them (s orbitals) in bonds where there is "more" electron density to stabilise. Since s orbitals are lower in energy than p orbitals, they are better at stabilising electrons. Before that, I'll note that we concern ourselves with the hybridisation of the orbitals at the central atom. What follows below is a crude explanation. 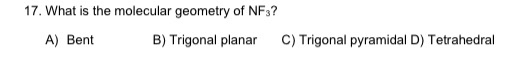
Electron pair arrangement molecular shape and ideal bond angles for NF3 trigonal planar trigonal planar 120 deg What is the correct molecular. It has three bond pairs and one lone pair. The compound NF3 is nitrogen trifluoride. with the fact that the ClF3 molecule has a dipole moment Justify your answer in terms of bond polarity and molecular structure. So the tetra hydra would be disrupted and we come in Tarragona phenomenal is nitrogen trifle right polar non polar is a polar substance because the distribution of charge due to this long pair is not a even so you have an uneven distribution of charge converting the substance in a polar molecule.This can be argued on the basis of Bent's rule concisely statedĪtomic s character concentrates in orbitals directed toward electropositive substituents Nitrogen trifluoride, with the chemical formula NF3, is an inorganic compound which is trigonal pyramidal in shape. We have that the electron geometry tetra hydro but the molecular geometry with the tribunal by Ramadan because we have by damming down Because we have four groups of electrodes but one of them is alone here. What is the electron geometry? We have four. We saw we would play the other two as a long pair on top of the sink. Although both the BF3 and NF3 molecules contain the same number of atoms. So we have to we would place them another seats to complete a for each of them. How is the structure around a given atom related to repulsion between valence. The shape of the molecule depends on how. here is the answer if anyone is intrested. Also, the dipole moment is a vector quantity and, therefore, directional. Study with Quizlet and memorize flashcards containing terms like Determine the electron geometry for each molecule CF4 NF3 OF2 H2S, Molecular geometry for. actually I got the answer from a friend, thanks if you did read this. So complete the all text for the outside atoms. Both the molecules have a trigonal pyramidal shape. Then place the outside arms around the central atom. CF4 NF3 OF2 H2S In which cases do you expect. 
This case is nitrogen, that is the least electro negative. Determine the electron geometry, molecular geometry, and idealized bond angles for each of the following molecules. Hence, the shape of NF3 is trigonal pyramidal, and the FNF bond angle is less than 10928. So in total, if we are then We have 26 valence electrons in total draw the lewis structures to draw the lewis structure First identify the central arm. There are three bond pairs and one lone pair on the central atom. And as we have three fluoride items we have Total of 21 valence electrons. So it has five valence electrons fluorine is in group 78. How many total balance of electrons valence electrons? That system has Nitrogen is in group five.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
